Online Database of Chemicals from Around the World
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Ningbo Wingoo Semiconductor Core Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wingooxc.com | |||
 | +86 18842662104 | |||
 | zoulin@wingooxc.com | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Chemical reagent >> Organic reagent >> Cyanate ester / isocyanate |
|---|---|
| Name | Monoallyldiglycidylisocyanuric acid |
| Synonyms | Allyl diglycidyl isocyanurate; Diglycidyl monoallyl isocyanurate; Monoallyl diglycidyl isocyanurate; Monoallyldiglycidylisocyanuric acid; NSC 329177 |
| Molecular Structure | 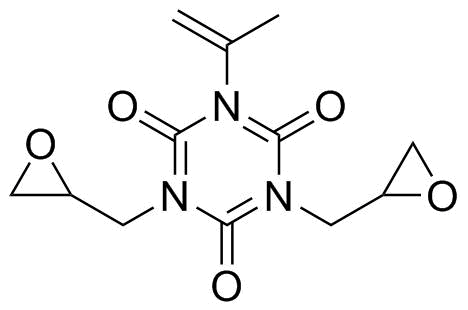 |
| Molecular Formula | C12H15N3O5 |
| Molecular Weight | 281.26 |
| CAS Registry Number | 69731-45-9 |
| EC Number | 819-936-3 |
| SMILES | O=C1N(C(=O)N(C(=O)N1CC2OC2)CC3OC3)CC=C |
| Solubility | Slightly soluble (2.81 g/L) (25 $degree$C), Calc.* |
|---|---|
| Density | 1.448$+/-$0.06 g/cm3 (20 $degree$C 760 Torr), Calc.* |
| Melting point | 48-49 $degree$C** |
| Boiling point | 448.3$+/-$31.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 224.9$+/-$24.8 $degree$C (Calc.)* |
| Index of refraction | 1.602 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 ($copyright$1994-2018 ACD/Labs) |
| ** | GB 941507 1963. |
| Hazard Classification | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||
|
Monoallyldiglycidylisocyanuric acid is a multifunctional organic derivative based on the isocyanuric acid (triazine-2,4,6-trione) core, in which three nitrogen atoms of the heterocyclic ring are selectively substituted with reactive functional groups: one allyl group and two glycidyl (epoxide-containing) groups. The resulting structure belongs to the class of multifunctional crosslinking monomers used in polymer and resin chemistry. The isocyanuric acid core is a six-membered heterocycle containing alternating carbonyl and nitrogen atoms (a 1,3,5-triazine-2,4,6-trione framework). This ring provides a rigid, thermally stable scaffold with three equivalent reactive nitrogen positions that can be functionalized. In monoallyldiglycidylisocyanuric acid, one of these nitrogen atoms is substituted with an allyl group (–CH2CH=CH2), while the remaining two are substituted with glycidyl groups containing epoxide rings. The allyl group introduces a carbon–carbon double bond that can participate in radical polymerization or thiol–ene addition reactions. The glycidyl groups contain strained three-membered epoxide rings, which are highly reactive toward nucleophiles such as amines, alcohols, and carboxylic acids. This combination of functionalities makes the molecule highly versatile as a multifunctional crosslinking agent. The isocyanuric acid derivatives of this type were developed as part of advances in epoxy resin and specialty polymer chemistry, where multifunctional monomers are used to increase crosslink density and improve thermal and mechanical properties of cured materials. By combining allyl and epoxide functionalities in a single molecule, it is possible to design systems that can undergo both radical and nucleophilic curing reactions, enabling flexible processing routes. Synthesis of monoallyldiglycidylisocyanuric acid typically involves stepwise substitution of isocyanuric acid or its derivatives. The process begins with selective alkylation or allylation of one nitrogen atom using allyl halides under basic conditions. The remaining nitrogen atoms are then reacted with epichlorohydrin in the presence of a base to introduce glycidyl (epoxy) groups. Careful control of reaction stoichiometry and conditions is required to achieve the desired substitution pattern and avoid over-functionalization. The presence of both allyl and epoxy groups gives the compound dual reactivity. The epoxide rings can undergo ring-opening polymerization or curing reactions with amine hardeners, forming crosslinked epoxy networks. The allyl group can participate in free-radical polymerization, allowing incorporation into radical-cured resin systems. This dual functionality makes the compound useful in hybrid polymer formulations. In polymer applications, monoallyldiglycidylisocyanuric acid is used as a reactive monomer or crosslinking agent in thermosetting resins, coatings, adhesives, and composite materials. The rigid triazine core contributes to thermal stability, while the multifunctional reactive groups increase crosslink density, resulting in materials with enhanced mechanical strength, chemical resistance, and heat resistance. The epoxide functionality is particularly important in epoxy resin chemistry, where curing with amines or anhydrides leads to three-dimensional network formation. The allyl group can also be used to tune curing kinetics or introduce additional crosslinking pathways through radical initiators or photochemical processes. From a structural standpoint, the molecule is highly functionalized and conformationally rigid due to the planar triazine ring. The distribution of reactive groups around this core allows for controlled network formation in polymer systems, making it valuable in advanced materials design. Overall, monoallyldiglycidylisocyanuric acid is a multifunctional triazine-based compound containing both allyl and glycidyl epoxy groups. Its combination of radical-reactive and nucleophile-reactive functionalities makes it an important crosslinking monomer in epoxy resins and specialty polymer systems, particularly where high-performance, thermally stable materials are required. References none |
| Market Analysis Reports |