Online Database of Chemicals from Around the World
| Nanjing Finetech Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fine-chemtech.com | |||
 | +86 (25) 5207-8417 +86 17714198479 | |||
 | +86 (25) 5207-8417 | |||
 | sales@fine-chemtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Ningbo Wingoo Semiconductor Core Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wingooxc.com | |||
 | +86 18842662104 | |||
 | zoulin@wingooxc.com | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Alcohols, phenols, phenolic compounds and derivatives |
|---|---|
| Name | 5,5'-Isopropylidenebis(m-xylene-2,a,a'-triol) |
| Synonyms | 4-[2-[4-hydroxy-3,5-bis(hydroxymethyl)phenyl]propan-2-yl]-2,6-bis(hydroxymethyl)phenol |
| Molecular Structure | 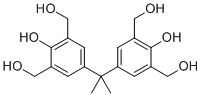 |
| Molecular Formula | C19H24O6 |
| Molecular Weight | 348.39 |
| CAS Registry Number | 3957-22-0 |
| EC Number | 223-553-1 |
| SMILES | CC(C)(C1=CC(=C(C(=C1)CO)O)CO)C2=CC(=C(C(=C2)CO)O)CO |
| Solubility | 1.142e+004 mg/L (25 $degree$C water) |
|---|---|
| Density | 1.4$+/-$0.1 g/cm3, Calc.* |
| Index of Refraction | 1.659, Calc.* |
| Melting point | 251.72 $degree$C |
| Boiling Point | 583.75 $degree$C, 564.8$+/-$45.0 $degree$C (760 mmHg), Calc.* |
| Flash Point | 261.4$+/-$23.3 $degree$C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P271-P280-P302-P304-P305-P313-P332-P337-P338-P340-P351-P352 Details |
| SDS | Available |
|
5,5'-Isopropylidenebis(m-xylene-2,a,a'-triol) is a polyphenolic organic compound belonging to the class of bisphenol-derived structures containing multiple hydroxyl functionalities. Its molecular architecture consists of two substituted aromatic units linked through an isopropylidene bridge (–C(CH3)2–), with each aromatic ring bearing several hydroxyl (–OH) groups and methyl substituents. This combination creates a highly functionalized, hydrogen-bonding-rich molecular framework. Structurally, the compound is based on a bisphenol-type core in which two aromatic rings are connected through a central carbon bearing two methyl groups. Each aromatic ring is further substituted with hydroxyl groups and methyl groups in a meta-xylene-like substitution pattern. The presence of multiple phenolic hydroxyl groups makes the molecule strongly polar and capable of extensive intermolecular hydrogen bonding, both within the molecule (intramolecular) and between molecules (intermolecular). The isopropylidene bridge is a key structural feature that imparts rigidity to the molecule. This bridge locks the two aromatic rings into a fixed spatial arrangement, reducing conformational flexibility. Such rigidity is often important in materials chemistry, where molecular shape influences thermal stability, glass transition behavior, and polymer-forming properties. Compounds of this general structural type are typically derived from condensation reactions involving phenolic precursors and carbonyl compounds such as acetone or related ketones. In classical bisphenol chemistry, acid-catalyzed condensation of phenols with acetone leads to isopropylidene-linked diphenolic structures. Further functionalization of the aromatic rings can introduce additional hydroxyl groups or alkyl substituents, yielding highly substituted polyphenolic derivatives. The hydroxyl-rich nature of 5,5'-isopropylidenebis(m-xylene-2,a,a'-triol) gives it strong hydrogen-bonding capability, which influences both its solubility and its potential for chemical reactivity. Phenolic hydroxyl groups can undergo typical aromatic alcohol reactions, including etherification, esterification, and oxidative coupling. These functional groups also make such compounds useful as intermediates in the synthesis of more complex organic structures. In a broader chemical context, highly substituted bisphenol derivatives are often studied for their role in polymer chemistry and resin formation. The presence of multiple reactive hydroxyl groups allows these molecules to act as multifunctional monomers or crosslinking agents in polymer networks. The isopropylidene linkage contributes rigidity, which can enhance thermal and mechanical properties in derived materials. Aromatic polyphenolic compounds of this type are also relevant in studies of antioxidant activity, as phenolic hydroxyl groups can donate hydrogen atoms to neutralize free radicals. This chemical behavior is characteristic of many phenol-rich structures, although specific activity depends on substitution patterns and molecular environment. From a structural chemistry perspective, the combination of aromatic rings, multiple hydroxyl substituents, and a rigid bridging group makes this compound highly polar and capable of complex intermolecular interactions. These features influence crystallization behavior, melting point, and potential compatibility with other polar or aromatic systems. Overall, 5,5'-isopropylidenebis(m-xylene-2,a,a'-triol) is a highly functionalized bisphenol-type compound characterized by a rigid isopropylidene bridge and multiple phenolic hydroxyl groups. Its structure places it within a broader class of polyphenolic molecules relevant to organic synthesis, hydrogen-bonding chemistry, and potential polymer-related applications. References The formation of ultralow-density microcellular diane-formaldehyde gels and aerogels. Polymer Science, Series B. DOI: 10.1134/s1560090416020081 |
| Market Analysis Reports |