Online Database of Chemicals from Around the World
| Ningbo Wingoo Semiconductor Core Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wingooxc.com | |||
 | +86 18842662104 | |||
 | zoulin@wingooxc.com | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Chemical reagent >> Organic reagent >> Azo, diazonium compound |
|---|---|
| Name | Bis(p-toluenesulfonyl)diazomethane |
| Molecular Structure | 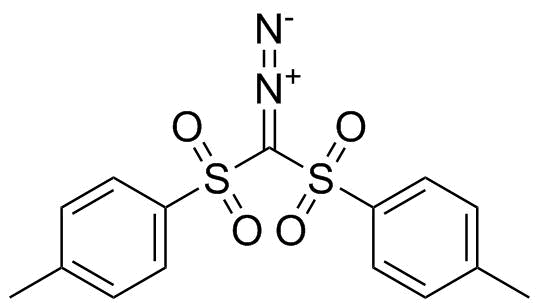 |
| Molecular Formula | C15H14N2O4S2 |
| Molecular Weight | 350.41 |
| CAS Registry Number | 14159-45-6 |
| SMILES | C1(=CC=C(C=C1)C)[S](C([S](C2=CC=C(C=C2)C)(=O)=O)=[N+]=[N-])(=O)=O |
|
Bis(p-toluenesulfonyl)diazomethane is a stabilized diazo compound in which a central diazomethyl unit (–CHN2) is flanked by two p-toluenesulfonyl (tosyl, –SO2–C6H4–CH3) groups. This structure places the reactive diazo functionality between two strongly electron-withdrawing sulfonyl substituents, which significantly enhances its stability compared with simple diazo compounds like diazomethane while still allowing controlled reactivity. The molecular structure consists of a central carbon bonded to a diazo group and two tosyl groups. The sulfonyl groups stabilize adjacent negative charge through resonance and inductive effects, making the compound a useful “soft” carbene precursor. The aromatic p-tolyl substituents also add steric bulk and improve crystallinity and handling properties. As a result, bis(p-toluenesulfonyl)diazomethane is often isolated as a relatively stable solid, though it remains a reactive species. Synthesis typically involves preparation of bis(p-toluenesulfonyl)methane, followed by diazo transfer using reagents such as sulfonyl azides under basic conditions. The reaction introduces the diazo functionality while preserving the sulfone groups. Careful control of temperature and purification conditions is required due to the sensitivity of diazo compounds to heat, light, and shock. This compound is widely used in organic synthesis as a carbene precursor. Under thermal or metal-catalyzed conditions (e.g., with rhodium or copper catalysts), it can generate a stabilized carbene species that participates in cyclopropanation of alkenes, insertion into C–H and heteroatom–H bonds, and ylide formation. Compared to unstabilized diazo reagents, it offers improved selectivity and safer handling. In addition, bis(p-toluenesulfonyl)diazomethane can serve as a methylene transfer reagent or participate in nucleophilic substitution processes after activation. The presence of the tosyl groups allows further functional transformations, including elimination or substitution to access diverse organic frameworks. Overall, bis(p-toluenesulfonyl)diazomethane is a valuable, stabilized diazo reagent that combines manageable handling with versatile reactivity. Its ability to generate controlled carbene intermediates makes it widely useful in modern organic synthesis, particularly for cyclopropanation and insertion reactions, while its sulfonyl groups provide both stabilization and synthetic flexibility. References none |
| Market Analysis Reports |