Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Sinyear Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinyear.com | |||
 | +86 (21) 5643-6307 | |||
 | +86 (21) 5168-5846 | |||
 | 42311713@qq.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Guangzhou Jun Xin Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jxinchem.com | |||
 | +86 (020) 8256-9208 | |||
 | +86 (020) 2826-9100 | |||
 | ivyyoung@gzjxchem.com | |||
 | QQ Chat | |||
 | WeChat: gzjx18122041046 | |||
 | WhatsApp:+8618122041046 | |||
| Chemical distributor since 2007 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty alcohol |
|---|---|
| Name | 2-Methyl-2,4-pentanediol |
| Synonyms | 2-Methylpentan-2,4-diol; Hexylene glycol |
| Molecular Structure | 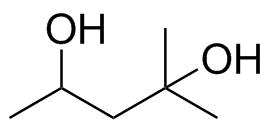 |
| Molecular Formula | C6H14O2 |
| Molecular Weight | 118.17 |
| CAS Registry Number | 107-41-5 |
| EC Number | 203-489-0 |
| SMILES | CC(CC(C)(C)O)O |
| Density | 1.0$+/-$0.1 g/cm3 Calc.*, 0.925 g/mL (Expl.) |
|---|---|
| Melting point | -40 $degree$C (Expl.) |
| Boiling point | 197.5 $degree$C 760 mmHg (Calc.)*, 197 $degree$C (Expl.) |
| Flash point | 93.9 $degree$C (Calc.)*, 94 $degree$C (Expl.) |
| Solubility | 10 mM in H2O (Expl.) |
| Index of refraction | 1.447 (Calc.)* |
| Alpha | 1.427 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | S26;S36 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
2-Methyl-2,4-pentanediol is an aliphatic diol with the molecular formula C6H14O2. It contains two hydroxyl groups located at the 2- and 4-positions of a branched pentane backbone, with an additional methyl substituent at the 2-position. This structure gives the molecule both hydrophilic and hydrophobic character, making it a versatile solvent and chemical intermediate. The molecular structure consists of a five-carbon chain (pentane) bearing hydroxyl groups at carbon 2 and carbon 4. The carbon at position 2 is also substituted with a methyl group, which increases steric bulk and influences the compound’s physical properties, such as viscosity and boiling point. The presence of two hydroxyl groups allows the molecule to form hydrogen bonds with water and other polar solvents, contributing to its high solubility in aqueous systems. 2-Methyl-2,4-pentanediol has been used in industrial and laboratory settings as a solvent and chemical intermediate. It is miscible with water and many organic solvents, which makes it useful in formulations requiring controlled polarity. Its relatively low volatility compared to simpler alcohols is due to hydrogen bonding between molecules, which increases intermolecular attraction and raises its boiling point. Historically, glycols and polyols such as 2-methyl-2,4-pentanediol became important in the development of industrial solvents and antifreeze agents in the 20th century. While ethylene glycol and propylene glycol are more widely known for antifreeze applications, branched diols like this compound have been studied for their unique balance of hydrophilicity, low freezing points, and solvent properties. In biochemical and pharmaceutical research, 2-methyl-2,4-pentanediol has been used as a precipitant and crystallization agent for proteins. Its ability to influence protein solubility and stabilize certain conformations makes it useful in structural biology, particularly in X-ray crystallography. It can promote controlled protein aggregation or crystal formation under carefully adjusted conditions, aiding in the determination of biomolecular structures. In industrial chemistry, the compound can serve as an intermediate in the synthesis of esters, ethers, and other functional derivatives. The hydroxyl groups can undergo esterification or etherification reactions, allowing incorporation into more complex molecules. Its branched structure can also influence the physical properties of resulting derivatives, such as flexibility, hydrophobicity, and thermal stability. The compound’s combination of moderate hydrophobic character and strong hydrogen bonding ability also makes it useful in coatings, inks, and formulation chemistry, where it can act as a co-solvent or plasticizing agent. Its ability to modify evaporation rates and solubility behavior is particularly valuable in systems requiring controlled drying or deposition. Overall, 2-methyl-2,4-pentanediol is a branched aliphatic diol with significant utility as a solvent, intermediate, and biochemical reagent. Its structure, featuring two hydroxyl groups and a methyl substituent, gives it a balance of polarity and hydrophobicity that underlies its applications in industrial chemistry, protein crystallization, and formulation science. References 2026. Conformation-selective detection of residues in solid proteins under magic-angle-spinning. Journal of Biomolecular NMR. DOI: 10.1007/s10858-025-00482-x 2026. Machine learning-assisted SERS detection of pyrethroid pesticides in edible fungi using a magnetic nanosensor. Microchimica Acta. DOI: 10.1007/s00604-025-07832-4 2026. Cross-species optimization of nuclei isolation in ten plant species. Plant Methods. DOI: 10.1186/s13007-025-01491-5 |
| Market Analysis Reports |